The major drawbacks of Dalton’s Atomic Theory are as follows:. Moreover, as scientists discovered in further research, though Dalton’s atomic theory was a significant turning point in the field of atomic theory, it had several flaws. Though Dalton’s Atomic Theory explained the concept of atoms in great detail, it was not without limitations. Masses and other Properties both (Chemical and Physical) are same in atoms of the same element and differ in the case of different elements. 
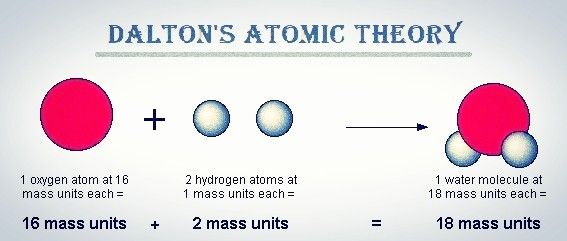
Reactant and compound are the outcomes of reshuffling of the atoms that already exists.This means atoms’ creation or destruction does not happen in a chemical reaction. When a chemical reaction happens, there are no atoms created or destroyed.Compounds are the result of combining atoms from distinct elements.This entails Atoms of same element = same mass whereas Atoms of different element = different mass And the masses and properties of atoms of different elements differ. The characteristics of the atoms of the same elements are the same, this is inclusive of mass.Atoms are particles that cannot be further divided and that constitute matter.It was founded on mass conservation and constant composition principles.Dalton’s atomic theory was the first comprehensive attempt to explain all matter in terms of atoms and their properties.It asserted that all matter was made up of small, indestructible particles known as ‘atoms.’.This theory was published in a paper titled “A New Chemical Philosophy” the philosophy was indeed novel at the time In 1808, John Dalton proposed the well-known Dalton’s Atomic Theory.This was called Dalton’s Atomic Theory.According to Dalton’s atomic theory, all substances are composed of atoms. At the beginning of the 19 th century, an English scientist named John Dalton presented the Atomic Theory that attempted to explain the composition of matter and formed the basis for the study of chemistry. Scientists have been trying to discover what composes matter for a long time.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
